Author: qp-audits
- Home
- Articles posted byqp-audits
qp-audits591 Comments
Navigating ANVISA’s Cleaning Validation Requirements — with a Spotlight on Solubility Criteria —
If you’re preparing for an ANVISA inspection—or simply raising the bar on
qp-audits50 Comments
FDA Updates NDSRI Implementation Timelines – What You Need to Know
On June 23, 2025, FDA quietly revised its “CDER Nitrosamine Impurity Acceptable
qp-audits34 Comments
The Future of Food Manufacturing: Advanced Techniques and Global Regulatory Developments
In an era where technology reshapes every industry, food manufacturing stands as
qp-audits43 Comments
Biotechnology Manufacturing: Innovation, Supply Chain Resilience, and Evolving Regulations
Biotechnology is no longer confined to beakers and test tubes—it’s now at
qp-audits69 Comments
Medication Therapy Management: Empowering Your Health Journey
In today’s fast-paced world, managing medications can feel overwhelming. Medication Therapy Management
qp-audits70 Comments
Implementing Industry 4.0: Embracing the Digital Revolution in Manufacturing
We’re on the brink of a digital renaissance—and it’s called Industry 4.0.
qp-audits73 Comments
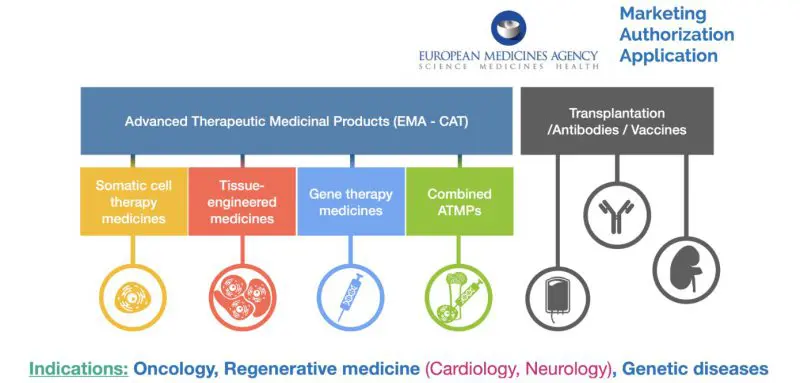
EMA’s New Wave: Transforming Human Medicinal Manufacturing
In today’s fast-paced healthcare landscape, ensuring that medicines meet the highest quality
qp-audits91 Comments
Biosimilars: A New Chapter in Accessible Healthcare
Biosimilars are reshaping the way we think about biologic medicines, bringing innovation
qp-audits98 Comments
New Regulatory Milestones in Veterinary Medicinal Products
The veterinary pharmaceutical sector is transforming rapidly, with regulatory changes that pave
qp-audits123 Comments
Streamlining Generic Drug Approvals: A Closer Look at the New ANDA Guidance
In June 2025, the U.S. Department of Health and Human Services and
Recent Posts
qp-audits50 Comments